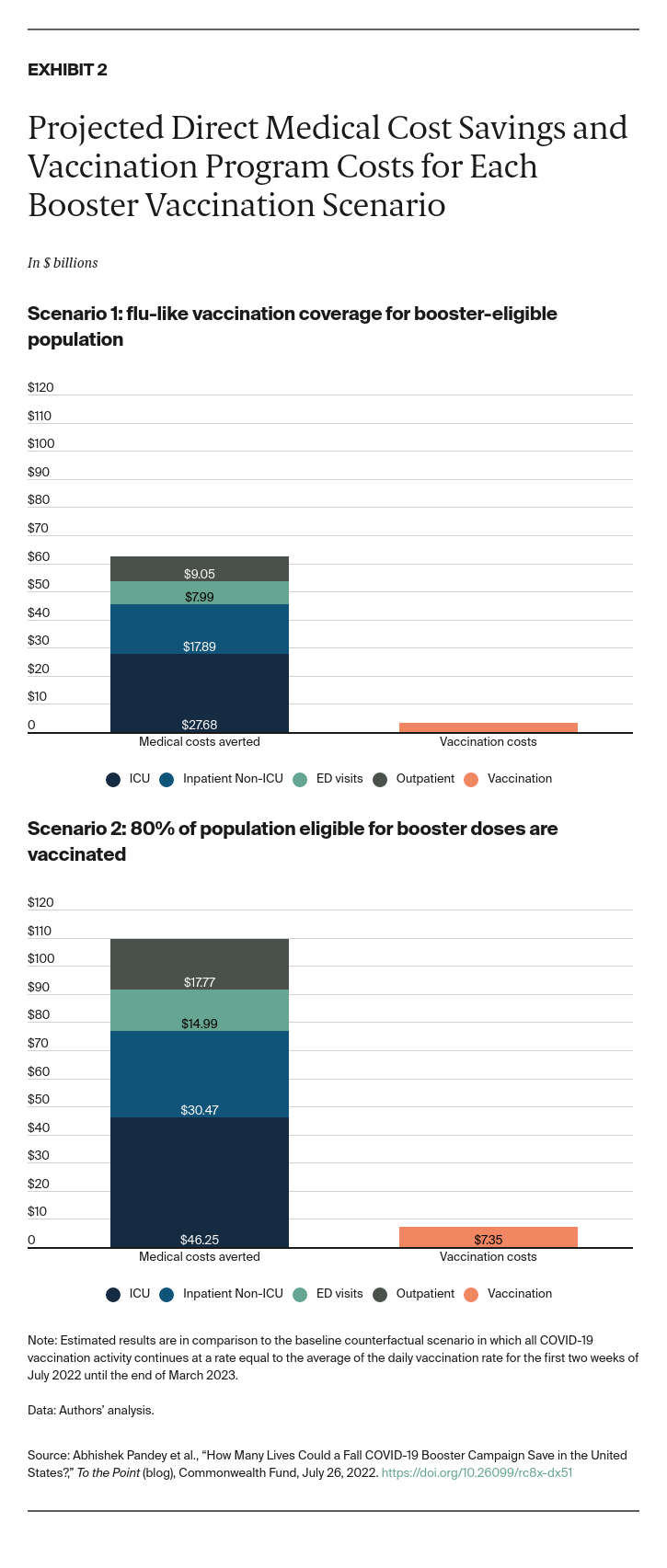
An early fall vaccination campaign could avert between $63 billion and $109 billion in medical costs, depending on level of booster coverage achieved, with the majority of savings resulting from averted hospitalizations, particularly in the ICU (Exhibit 2). Vaccine doses and administration costs would rise between $3.2 billion and $7.4 billion compared with the baseline scenario where vaccination rates remain at the current pace. At booster coverage mirroring the 2020–2021 influenza campaign, this translates to $1,241 in savings per dose. In moving from the lower coverage target to the higher, savings of $706 per dose would be realized.
Need for Ongoing Vaccination Funding
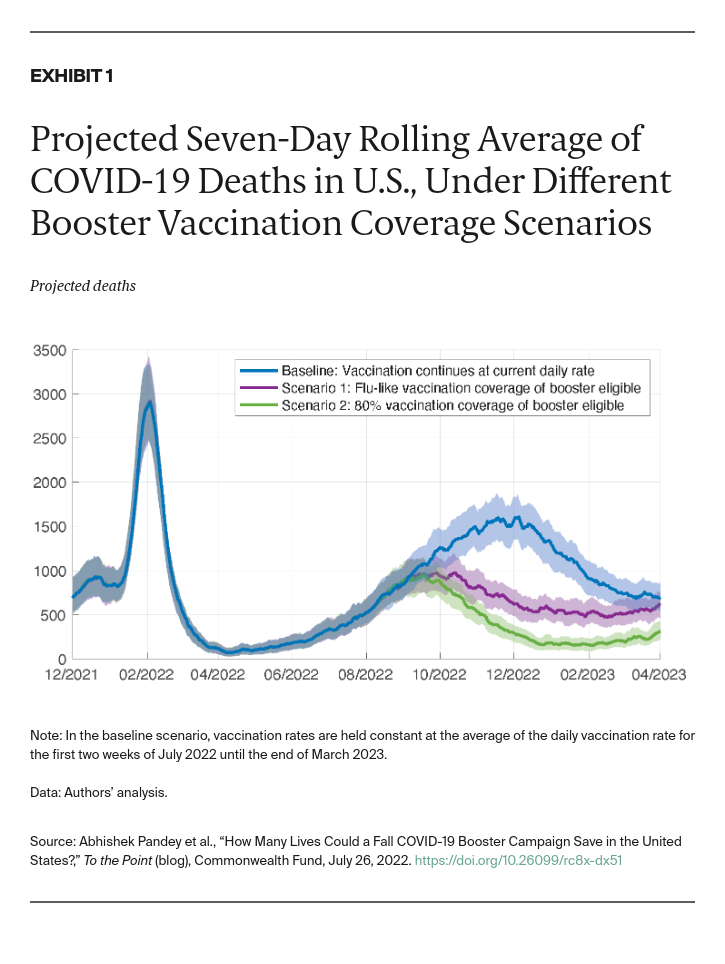
As population immunity wanes and new SARS-CoV-2 variants capable of evading vaccine protection emerge, an uptick in hospitalizations and deaths seems increasingly likely. Congress has failed to authorize additional funding for COVID-19 vaccines and treatments, hindering access at a crucial time. The Biden administration has not yet expanded eligibility for a second booster for all Americans who have received their first booster, but has considered doing so. A fall vaccination campaign that expands eligibility for boosters and moves aggressively to reach people could avert a surge of hospitalizations and deaths peaks in December. Our results indicate that a such an increase in late 2022 could lead to more than 2.7 million hospitalizations and more than 260,000 deaths by the end of March 2023 if vaccination continues at its current rate.
A campaign that mirrors seasonal influenza vaccination would save both lives and costs. If the COVID-19 vaccination campaign coincides with seasonal flu shots, public health officials could use existing outreach and delivery mechanisms to achieve vaccination targets, reducing costs even further than we estimate here. A fraction of the cost savings generated could be earmarked for public health outreach dedicated to vaccination.
Our analysis was conducted with current knowledge of vaccine effectiveness against the Omicron subvariants. Although we considered waning vaccine-induced and naturally acquired immunity, our estimates assume that currently available vaccines will continue to roll out and that the virus evolution does not lead to the emergence of significantly different variants. If Omicron-specific vaccines with improved effectiveness are administered, our results may underestimate vaccine impact. The Food and Drug Administration may soon review data for new vaccines that more closely match circulating variants. With more efficacious vaccines, a fall campaign would suppress COVID-19 infections, hospitalizations, and deaths even further.
While current vaccines still provide considerable protection against severe illness and death, they are less effective in preventing infections from the subvariants of Omicron such as BA.4 and BA.5. Increasingly rapid spread of these subvariants among the U.S. population, along with relaxing mandates, could lead to a wave that arrives earlier and is much larger in magnitude than what we have predicted in our analysis. As such, it will be important to ramp up booster vaccination as early as possible.
The continued absence of new federal funding for COVID-19 vaccination will limit efforts to increase booster vaccination coverage and could lead to thousands of avoidable hospitalizations and deaths.