Introduction
There is a maternal health crisis in the United States. The number of pregnant and birthing people dying more than doubled between 1987 and 2018, rising from 7.2 deaths per 100,000 live births to 17.4 deaths per 100,000 live births.1 Moreover, racial disparities are stark: Black pregnant and birthing people are three times more likely to die from pregnancy-related causes than their white counterparts; Indigenous Americans are approximately twice as likely.2 Geographic disparities are also evident, with rural Americans experiencing rates of maternal mortality twice as high as those for urban residents.3
But maternal mortality is only the tip of the iceberg. Rates of severe maternal morbidity (SMM), which includes unexpected, life-threatening outcomes (often called “near deaths”), are also rising. The latest estimates indicate that more than 60,000 birthing people per year experience severe maternal morbidity, with similarly wide racial and geographic disparities as those found with maternal mortality.4
As bipartisan legislation in the U.S. House of Representatives and the Senate seeks to address the maternal health crisis and narrow racial disparities in outcomes, this report is intended to inform policy aimed at lowering rates of maternal morbidity and mortality and eliminating inequities. We highlight both the evidence underpinning various approaches and the importance of employing an equity framework in designing and implementing these policies.
Given the rapid evolution of the field of maternal health and the large body of research available, we have sought to describe the most recent and relevant findings. Nevertheless, our report is not intended as a comprehensive systematic review of the literature. We compiled evidence based on searches of the peer-reviewed and gray literature, review of references, and consultation with clinical and research expertise in pregnancy and maternal health.
Much of our research was undertaken prior to the start of the COVID-19 pandemic. New evidence is emerging about how COVID-19 has exacerbated many of the adverse outcomes and inequities we describe, thus heightening the urgency of action on maternal mortality and morbidity.
What the Available Evidence Shows
Making Critical Investments to Address Social Determinants of Health
Social determinants of health (SDOH) are defined as “conditions in environments in which people are born, live, learn, work, play, worship, and age that affect a wide range of health, functioning, and quality-of-life outcomes and risks” (Exhibit 1).5 Examples include housing security, convenient access to nutritious food, employment opportunities, and the safety of the home or neighborhood environment. SDOH have long been recognized as important factors in maternal morbidity and mortality.6
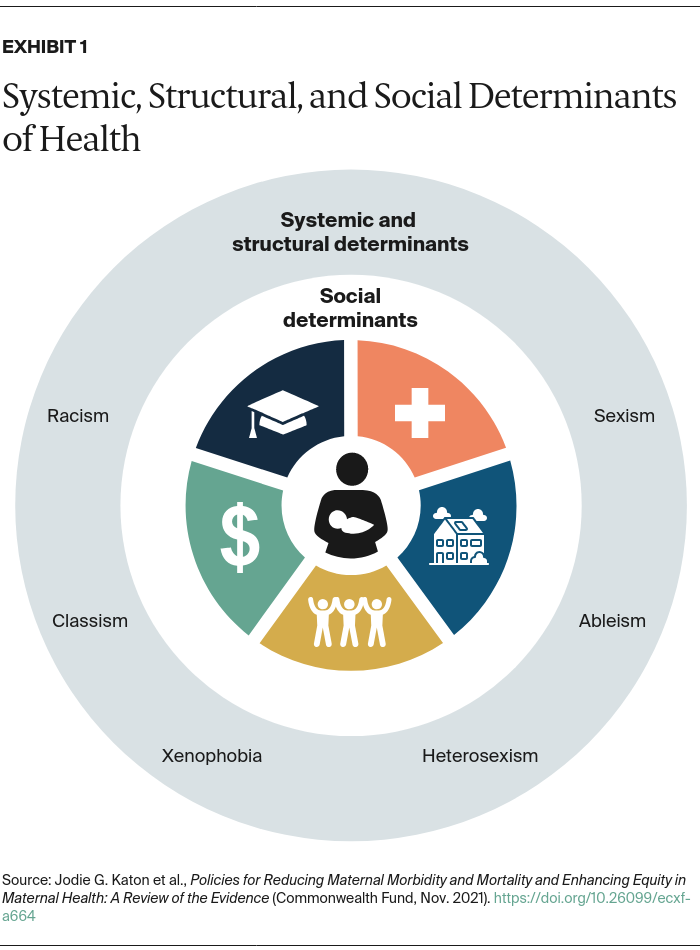
In the U.S., SDOH disproportionately affect people of color, owing to systemic racism and policies dating back to the country’s founding that have been directly or indirectly discriminatory in their design and/or implementation. In turn, these policies have led to pronounced racial inequities in access to quality health care and education, food security, safe housing, employment, and more, ultimately producing racial disparities in health outcomes.7
Many SDOH are interrelated, and they can affect pregnancy outcomes through complex interactions.8 For example, prepregnancy health and health behaviors that increase risk for maternal morbidity and mortality, such as hypertension and lack of physical activity, are influenced by the availability of safe to places to exercise and access to affordable, nutritious food. The health of women is further undermined when SDOH result in financial insecurity, with the related stress often resulting in compromised physical and mental health.9 Additional studies of maternal health point to the impact of racism on stress, health, and well-being.10
Economic security. The systemic and structural factors that underpin SDOH have led to lower incomes and disparate educational and employment opportunities for people of color. Economic insecurity, in turn, has contributed to a higher burden of preventable health conditions in communities of color.11 Understanding racial disparities in maternal morbidity and mortality requires connecting these outcomes with their economic foundations.
For instance, the median white family in the U.S. holds nearly 10 times the wealth of the median Black family.12 A major cause of the wealth gap is historical redlining, the discriminatory practice of restricting access to financing and economic opportunities that helped to shape highly segregated communities across the country. Broadly, lower levels of wealth and income and higher levels of poverty are associated with greater risks of morbidity and mortality.13 Racial disparities in maternal morbidity and mortality, however, are apparent across all income and educational levels.14 For example, one study in New York City found that even among those living in the highest-income communities, Black pregnant and birthing people had a maternal mortality ratio nearly four times that for non-Black pregnant and birthing people.15 This suggests the need for equitable policy solutions that address not only SDOH but also structural racism overall and bias in health care settings.16
Neighborhood and built environment. In the U.S., where a person lives and the quality of their physical environment reflect historical and current discriminatory housing and loan practices. Such practices have led to continued segregation, with lower housing quality, environmental hazards, and other risks to health and safety concentrated in neighborhoods primarily populated by people of color. Housing instability and poor housing quality are associated with poorer maternal mental health, which in turn is a risk factor for maternal morbidity and mortality.17 There is growing evidence of the potential impact of environmental contaminants and pollution on risk of pregnancy complications such as preeclampsia and placental abruption, which in turn can raise the risk of maternal morbidity and mortality.18
Social and community context. Living in an unsafe community or social context can have physical and mental health consequences. Because of the legacy of redlining and continued housing segregation, pregnant and birthing people of color are more likely than white pregnant or birthing people to reside in communities with higher rates of crime, interpersonal violence, instability, and overpolicing.19
Living in a community with high rates of violence, including police violence and mass incarceration, is associated with both direct and indirect increased risk of maternal morbidity and mortality and preterm birth.20 Evidence highlights the profound negative impacts of interpersonal violence on maternal morbidity and mortality, including increased risk of pregnancy-associated homicide and suicide as well as other maternal mental health issues.21 Extensive research has clarified the role of neighborhood deprivation, crime rates, racial segregation, and other characteristics on birth outcomes, specifically preterm birth and low birth weight.22 However, there is significantly less research into how these factors affect maternal outcomes such as chronic health conditions and severe maternal morbidity.
Health care access. Access to health care is interwoven with both economic stability and neighborhood environment. Black people tend to give birth in hospitals marked by lower quality-of-care indicators than the hospitals where white people give birth.23 One study suggests that patient- and hospital-level factors — for example, the proportion of patients who are Medicaid beneficiaries — may be more immediate determinants of maternal morbidity than neighborhood factors like median household income.24 Compared to urban dwellers, rural residents have a 9 percent higher risk of maternal morbidity and mortality, with lack of access to obstetric care likely a key contributor to this disparity.25
The Affordable Care Act (ACA) has been particularly important in ensuring greater access to and use of health services before and after pregnancy.26 The law’s expansion of Medicaid eligibility appears to have been particularly effective in slowing the rise of maternal mortality among Black pregnant and birthing people in states that have taken up the expansion.27 The ACA also mandated coverage of preventive care, including contraception, and prohibited discrimination based on preexisting conditions, including pregnancy.
Growing and Diversifying the Perinatal Workforce
Nearly all U.S. births (98%) occur in hospital settings.28 Approximately 91 percent of hospital births are attended by a physician, while 8.7 percent are attended by midwives. This is unique to the United States; the majority of other high-income countries rely much more heavily on midwifery care and have fewer hospital births.29 Access to midwifery care is currently limited by state “scope of practice” laws, which in many states require nurse midwives to be supervised by a physician.30 Lack of coverage via Medicaid or many commercial insurers and high out-of-pocket costs also prevent access to doula services and nonhospital births.
Barriers to access particularly affect low-income and birthing people of color, who may get greater benefit from access to these services.31 Midwifery and doula care can raise patient satisfaction, reduce medical interventions during childbirth, and improve maternal and neonatal morbidity and mortality, particularly among women at higher risk of these outcomes.32
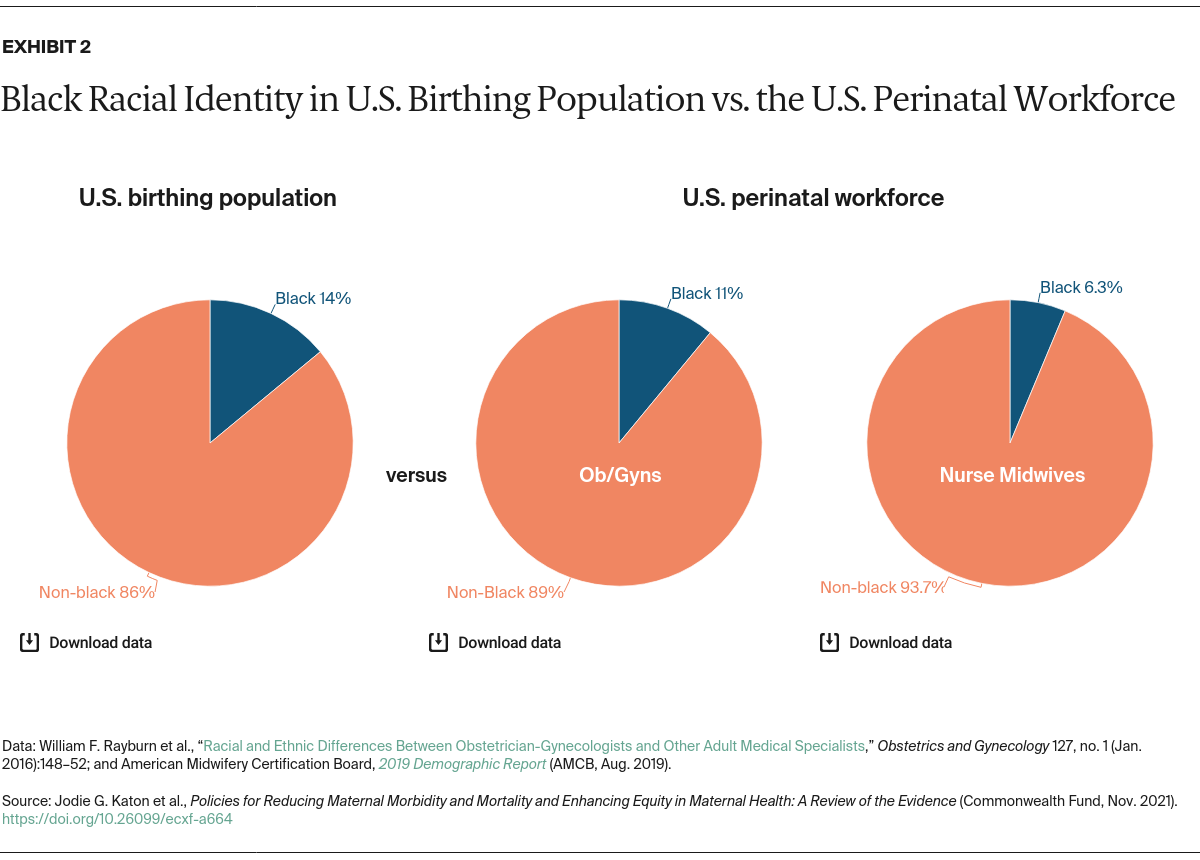
Not only is the birth workforce lacking in diversity in terms of training and discipline, but it does not reflect the racial and ethnic identities of the birthing population in the U.S. For example, while 14 percent of birthing individuals in the U.S. identify as Black, only 6.3 percent of certified nurse midwives and 11 percent of ob-gyns identify as Black (Exhibit 2).33 The high cost for enrolling and completing doula and midwifery training, low payment once in the field, and racism within training programs serve as barriers for people of color entering the perinatal workforce.34